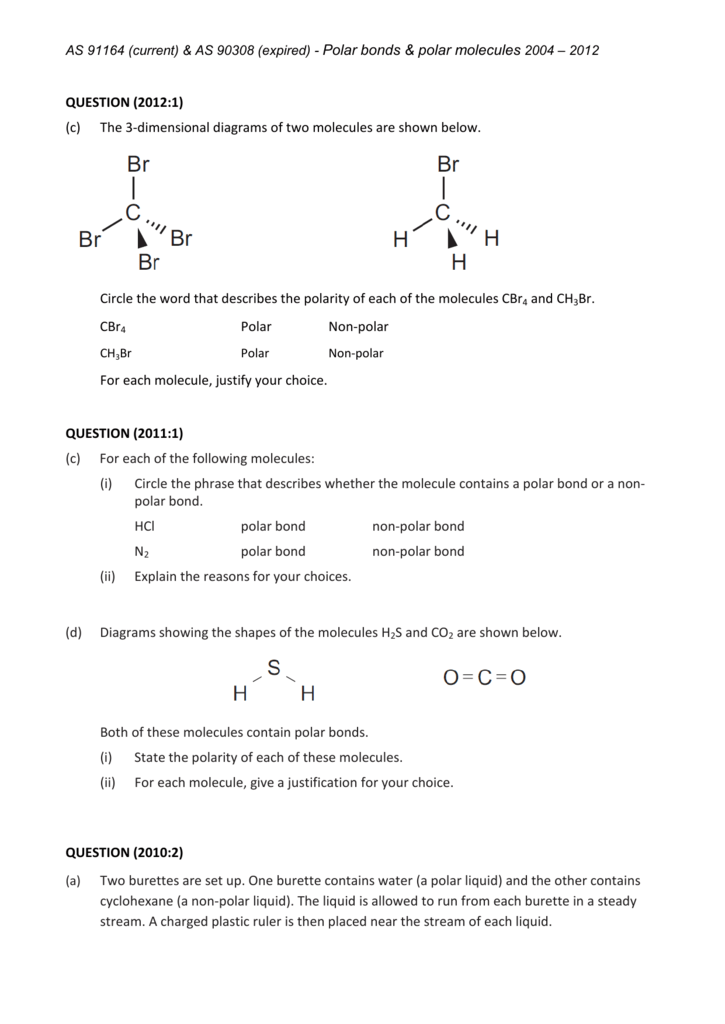
CH Br Polar Nonpolar
Properties of Carbon tetrabromide It has a molar mass of 331.627 g·mol −1. It is insoluble in water. It has a boiling point of 189.7 °C and a melting point of 94.5 °C. It is denser than water. It has a monoclinic tetrahedral structure. Page Contents show How to draw lewis structure for CBr4?

Cbr4 Electron Dot Structure
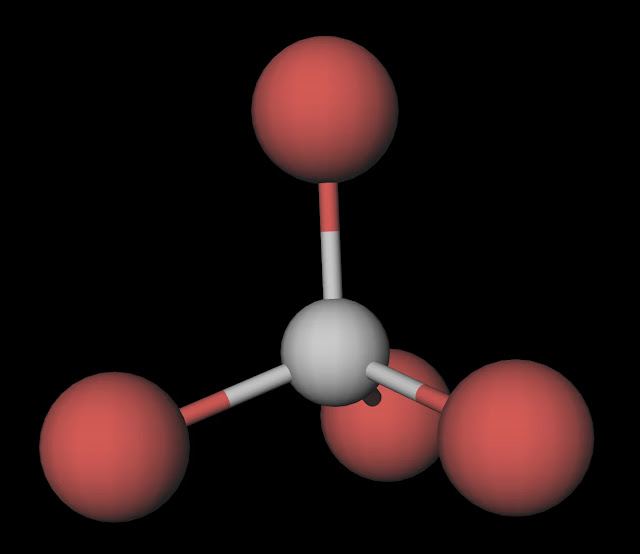
Carbon tetrabromide (CBr4) is a non-polar molecule. The central carbon (C) atom in the CBr4 molecule is surrounded by four bromine (Br) atoms via single covalent bonds, forming a symmetric tetrahedral molecule. The electronegativity of the bromine (Br) atom is slightly more than the carbon (C) atom.

The carbon tetrabromide molecule, CBr4, is A a polar molecule with
Henry Agnew (UC Davis) 5.10: Electronegativity and Bond Polarity is shared under a not declared license and was authored, remixed, and/or curated by LibreTexts. Covalent bonds can be nonpolar or polar, depending on the electronegativities of the atoms involved. Covalent bonds can be broken if energy is added to a molecule.
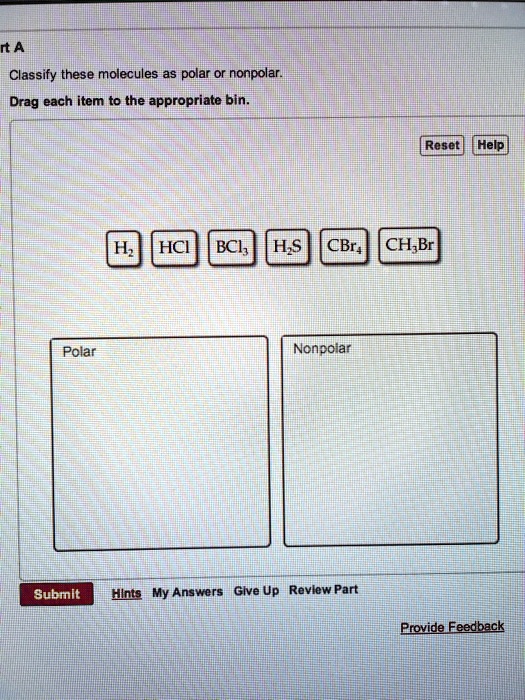
SOLVED rtANClassify these molecules as polar or nonpolar Drag each
Hello Everyone!Do you want to find out if Carbon Tetrabromide is a polar or nonpolar molecule? If yes then check out this video where we share our detailed s.
Cbr4 Lewis Structure
Why is a C-Br bond considered polar? From electronegativity considerations, both carbon and bromine have very similar electronegativities - 2.5 and 2.8 - respectively. Nonetheless, I am told that such a bond would be polar. We generally classify bond between atoms with EN differences < 0.5 as non-polar; why would C-Br be an exception? bond polarity
Solved 13) Choose the most polar molecule a) CH3Br b) CBr4
Carbon tetrabromide Carbon tetrabromide, CBr 4, also known as tetrabromomethane, is a bromide of carbon. Both names are acceptable under IUPAC nomenclature . Production CBr 4 can be obtained by the bromination of methane. The byproducts include other brominated methanes ( methyl bromide, dibromomethane and bromoform) and hydrogen bromide.

Cbr4 Electron Dot Structure
The CBr4 molecule is non-polar due to its symmetrical tetrahedral geometry. The four bromine atoms are arranged symmetrically around the central carbon atom, with each bond angle at 109.5 degrees. This results in an even distribution of charge throughout the molecule, with no net dipole moment.

Nama Senyawa Cbr4 Brain
The CBr4 molecule is non-polar.. Both CBr4 and CH3Br have four regions of electrons around the central carbon atom. These are all bonding electron regions (clouds) so the shape of both molecules is tetrahedral. The C-Br bond is polar due to the difference in electronegativity between C and Br. Is carbon polar and nonpolar?
Cbr4 Electron Dot Structure
Learn to determine if CBr4 is polar or nonpolar based on the Lewis Structure and the molecular geometry (shape).We start with the Lewis Structure and then us.
MakeTheBrainHappy Is CBr4 Polar or Nonpolar?
The molecule CBr4 is non-polar. This is because the charges from the bromine atoms cancel out, resulting a neutral charge. The molecule CBr is called tetrabromomethane, but is commonly known as carbon bromine.

Is CBr4 Polar or Nonpolar? Techiescientist
The C-O bond is considerably polar. Although C and S have very similar electronegativity values, S is slightly more electronegative than C, and so the C-S bond is just slightly polar. Because oxygen is more electronegative than sulfur, the oxygen end of the molecule is the negative end. Chloromethane, CH 3 Cl, is another example of a polar.

Is CBr4 Polar or Nonpolar? Techiescientist
When you place a molecule with an electric dipole in an electric field, a force acts to turn the molecule so that the positive and negative ends line up with the field. The magnitude of the turning force is given by the formula. µ = q × d. where q is the amount of charge and d is the distance between the two charges. µ is the turning moment.
Ch4 Polar Or Nonpolar Methane Ch4 Polar Or Nonpolar / Is Ch4 Polar Or
Other names: Methane, tetrabromo-; Carbon bromide (CBr4); Methane tetrabromide; Tetrabromomethane; CBr4; Carbon bromide; Bromid uhlicity; UN 2516; NSC 6179 Permanent link for this species. Use this link for bookmarking this species for future reference. Information on this page: Reaction thermochemistry data; Mass spectrum (electron ionization)

Ch4 Polar Or Nonpolar / Solution Is The Ch4 A Polar Or Non Polar Chemistry
Other names: Methane, tetrabromo-; Carbon bromide (CBr4); Methane tetrabromide; Tetrabromomethane; CBr4; Carbon bromide; Bromid uhlicity; UN 2516; NSC 6179 Permanent link for this species. Use this link for bookmarking this species for future reference. Information on this page: Notes; Other data available: Gas phase thermochemistry data
Cbr4 Electron Dot Structure
CBr4 (Carbon tetrabromide) is nonpolar in nature because of the symmetrical arrangement of four bromine atoms around carbon. As a result, the dipoles of the C-Br bond get canceled by each other resulting in CBr4 a nonpolar molecule.

Cbr4 Electron Dot Structure
May 27, 2023 by Jay Rana CBr4 is a covalent (nonpolar covalent) compound because when one nonmetal combines with another nonmetal, it usually forms a covalent compound. Here, C is a nonmetal and Br is also a nonmetal. So when they combine, it forms a covalent compound.